노록시코돈
Noroxycodone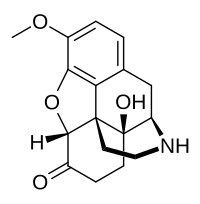 | |
| 식별자 | |
|---|---|
| |
| CAS 번호 | |
| 펍켐 CID | |
| 켐스파이더 | |
| 유니 | |
| CompTox 대시보드 (EPA) | |
| ECHA InfoCard | 100.055.334 |
| 화학 및 물리적 데이터 | |
| 공식 | C17H19NO4 |
| 어금질량 | 301.342 g·190−1 |
| 3D 모델(JSmol) | |
| |
| |
노록시코돈은 오피오이드 진통제 옥시코돈의 주요 대사물이다.[1][2][3]그것은 주로 CYP3A4에 의해 N-데메틸화를 통해 간에 있는 옥시코돈으로부터 형성된다.[1][2][3]노록시코돈은 옥시코돈과 유사한 μ-오피오이드 수용체(MOR)를 결합하여 활성화하지만 옥시코돈의 친화력은 1/3이고 활성도는 5~10배 낮다.[1][4][5]그러나 강력한 MOR 작용제임에도 불구하고 노록시코돈은 혈뇌-뇌-배리어와 중앙 신경계를 잘 교차하지 못하며, 이러한 이유로 인해 그에 비해 진통제는 극히 적다.[6][5][4][7]
참고 항목
참조
- ^ a b c Smith H, Passik S (25 April 2008). Pain and Chemical Dependency. Oxford University Press, USA. pp. 195–. ISBN 978-0-19-530055-0.
- ^ a b McPherson RA, Pincus MR (31 March 2016). Henry's Clinical Diagnosis and Management by Laboratory Methods. Elsevier Health Sciences. pp. 336–. ISBN 978-0-323-41315-2.
- ^ a b Anzenbacher P, Zanger UM (29 May 2012). Metabolism of Drugs and Other Xenobiotics. John Wiley & Sons. pp. 420–. ISBN 978-3-527-32903-8.
- ^ a b Lemberg KK, Siiskonen AO, Kontinen VK, Yli-Kauhaluoma JT, Kalso EA (February 2008). "Pharmacological characterization of noroxymorphone as a new opioid for spinal analgesia". Anesthesia and Analgesia. 106 (2): 463–70, table of contents. doi:10.1213/ane.0b013e3181605a15. PMID 18227301.
- ^ a b Preedy VR (25 April 2016). Neuropathology of Drug Addictions and Substance Misuse Volume 3: General Processes and Mechanisms, Prescription Medications, Caffeine and Areca, Polydrug Misuse, Emerging Addictions and Non-Drug Addictions. Elsevier Science. pp. 462–464. ISBN 978-0-12-800677-1.
- ^ Lalovic B, Kharasch E, Hoffer C, Risler L, Liu-Chen LY, Shen DD (May 2006). "Pharmacokinetics and pharmacodynamics of oral oxycodone in healthy human subjects: role of circulating active metabolites". Clinical Pharmacology and Therapeutics. 79 (5): 461–79. doi:10.1016/j.clpt.2006.01.009. PMID 16678548.
- ^ Klimas R, Witticke D, El Fallah S, Mikus G (May 2013). "Contribution of oxycodone and its metabolites to the overall analgesic effect after oxycodone administration". Expert Opinion on Drug Metabolism & Toxicology. 9 (5): 517–28. doi:10.1517/17425255.2013.779669. PMID 23488585.


