오달라스비르
Odalasvir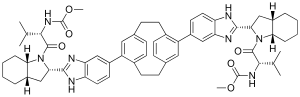 | |
| 법적현황 | |
|---|---|
| 법적현황 |
|
| 식별자 | |
| |
| CAS 번호 | |
| 펍켐 CID | |
| 켐스파이더 | |
| 유니 | |
| 화학 및 물리적 데이터 | |
| 공식 | C60H72N8O6 |
| 어금질량 | 1001.286 g·190−1 |
| 3D 모델(JSmol) | |
| |
| |
오달라스비르([2][3]INN,[1] 이전에 ACH-3102)는 C형 간염 치료를 위해 개발 중인 조사 신약이다.[4]그것은 NS5A 억제제다.[5]NS5A 단백질은 바이러스 복제를 포함한 바이러스성 수명 주기의 다양한 단계에서 다양한 기능을 제공한다.NS5A는 또한 치료 실패의 공통원인 인터페론 저항의 개발에도 역할을 한다.[6][7][8]그것은 Achillion Pharmetics에 의해 개발되고 있다.
참고 항목
참조
- ^ "International Nonproprietary Names for Pharmaceutical Substances (INN). RECOMMENDED International Nonproprietary Names: List 73" (PDF). who.int. World Health Organization. p. 38. Retrieved 5 December 2015.
- ^ "Achillion Has Discovered and Developed a Comprehensive Portfolio of Antivirals for the Treatment of Hepatitis C". Achillion Pharmaceuticals. Retrieved 15 November 2015.
- ^ "Odalasvir". ChemIDplus.
- ^ Walker T (December 2015). "Watch list 2016: top therapeutic areas: experts say you should follow these 6 therapeutic areas". Managed Healthcare Executive: 47. Retrieved March 12, 2016.
- ^ Nakamoto S, Kanda T, Wu S, Shirasawa H, Yokosuka O (March 2014). "Hepatitis C virus NS5A inhibitors and drug resistance mutations". World Journal of Gastroenterology. 20 (11): 2902–12. doi:10.3748/wjg.v20.i11.2902. PMC 3961994. PMID 24659881.
- ^ "ACH-3102 resources". Achillion Pharmaceuticals. Winter 2011. Retrieved 1 May 2012.
- ^ "Achillion gets FDA incentives for hepatitis C drug". Associated Press. 15 May 2012.
- ^ Levin J (18–22 April 2012). Preclinical characteristics of ACH-3102. 47th Annual Meeting. Barcelona, Spain: European Association for the Study of the Liver.


